|
The shape and size of the particle decide the velocity, with which particles migrate towards the electrode. When the pH of the solvent is below pH, the protein will show a positive charge and migrate towards the cathode. The protein exhibits a net negative charge when the pH of the solvent is above pI and migrates towards the anode. At the isoelectric point (pI) of a particular protein, the net charge in the protein is zero and it does not migrate in the electric field. If there are excesses of negative charges, protein will move faster towards the anode. If there are more positive charges, protein will migrate faster to the cathode. The direction and speed of protein migration are influenced by buffer pH.Īs a function of the environment, protein’s net charge is determined as the sum of the electric charges present on the surface. The pH of the solvent is maintained by the usage of buffers at different pH. 
Mobility or movement or migration is directly proportional to the magnitude of charge possessed, which is dependent on the solvent’s pH. Mobility is the ratio between the velocity of the ion and electric field strength. If there is a zwitterion, the overall charge is neutral, which will lead to zero mobility. The distance migrated is directly proportional to the time of passage of the electric field.īased on the pH of the solvent, the particle carries charges since it is amphoteric. 
Voltage is the force, which drives the molecule towards the electrodes. When a potential difference is applied across the electrodes, potential gradient is generated, which is the ratio of voltage and distance between the electrodes. Starch gel, cellulose acetate gel, polyacrylamide gel, and agarose gel are used as supporting media.ĭepending on the net charge carried out by the molecule in the presence of an electric field, the molecule migrates towards anode or cathode. The separation of molecules is dependent on the ratio of charge to mass of protein, pore size of medium and molecular size.Supporting medium is a matrix (gel), in which separation of biomolecules takes place.They also determine the thickness of the cloud of ions. Thus buffers determine extent of solute ionization, charge on solute and direction of migration.Setting of pH at which process is carried out.The external environment consists of the pH of the solution, the electric field applied the viscosity of temperature and temperature. Inherent factors are the magnitude of charge, charge density, molecular weight, and quaternary or tertiary structure (shape). Electrophoretic velocity depends on both inherent factors and the external environment.The rate of migration of biomolecules depend on the following factors: There are numerous factors which impact the electrophoresis process. 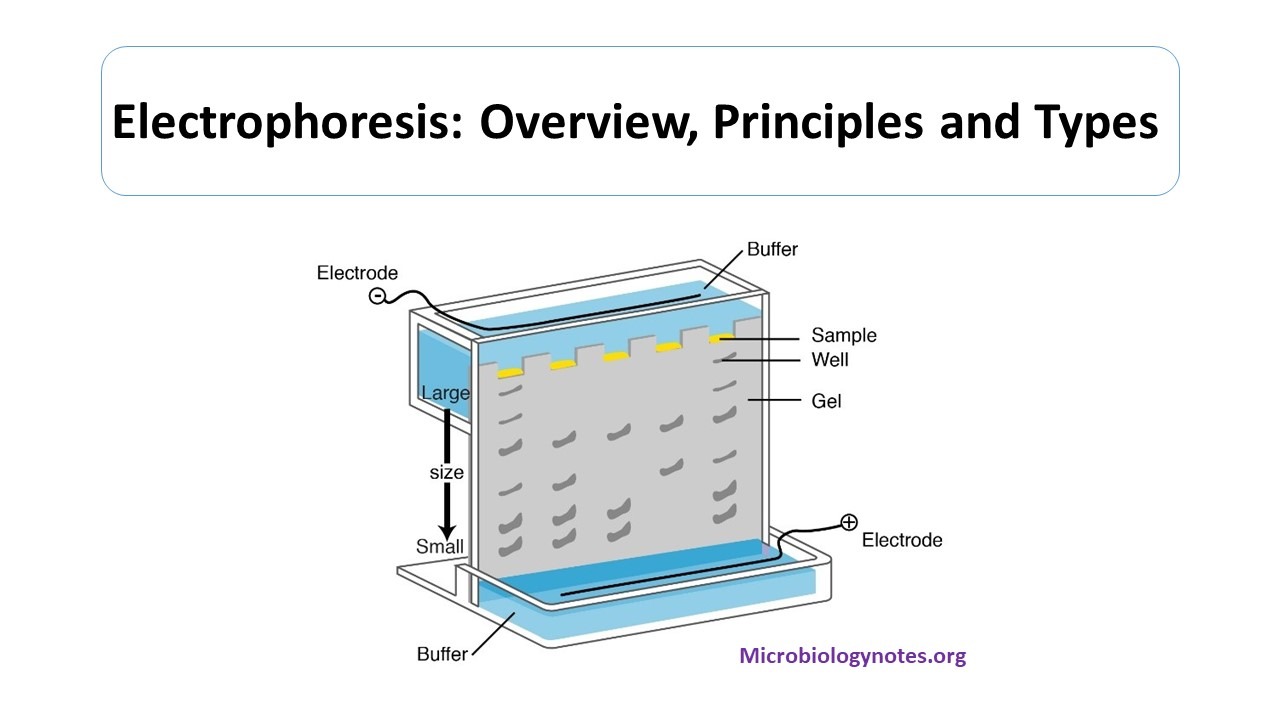
Some electrophoresis processes are based on the charge of molecules, while some rely on the molecular size of molecules. But, protein will carry a negative or positive charge depending on solvent pH and isoelectric point.Īn electrophoresis is an incomplete form of electrolysis because the electric field is removed before the molecules reach the electrode, yet the molecules have already been separated due to electrophoretic mobilities Then, the separated molecules are located with staining by a dye or autoradiography (for radioactive sample). Nucleic acid has a negative charge due to the phosphate backbone and so, it migrates towards the anode in DNA electrophoresis. Ions moving towards cathode are cations and those moving towards anode are anions. For eg- Protein consists of ionizable carboxyl and amino groups. Also, the ampholyte gets a positive charge in acidic conditions and moves towards the cathode. Ampholyte becomes negatively charged in basic conditions and migrates towards the anode. Depending on the charge carried by molecules, they move either towards anode or cathode. Under the influence of the electric field, the migration of charged particles happens in the liquid medium.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
